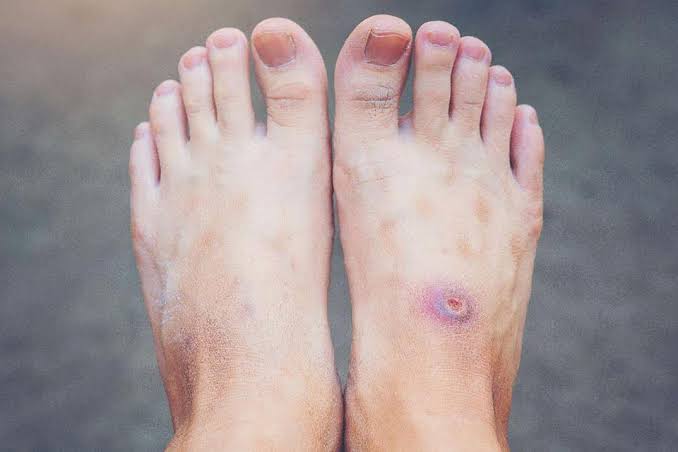
Why do diabetic wounds refuse to heal? A new scientific review highlights how disruptions in the timing and behavior of immune cells may hold the key to understanding this widespread medical challenge.
Chronic diabetic ulcers are among the most serious and costly complications associated with diabetes. More than 131 million people around the world are affected, and treatment costs reach an estimated $755 billion each year. These wounds often lead to amputation and carry a high risk of death, highlighting the urgent need to better understand the biological processes that prevent them from healing.
The immune system plays a central role in coordinating wound repair. However, many studies have not fully examined how different immune cell populations change over time in diabetic wounds. A comprehensive review led by Yi Ru and colleagues addresses this gap.
The researchers systematically analyzed how a wide range of immune cells behave during the different stages of diabetic wound healing.
The review examines immune cells involved in wound healing and how diabetes disrupts their functions, with particular focus on monocytes and macrophages.
Normally, monocytes become macrophages that shift from a pro-inflammatory M1 state to a pro-repair M2 state, helping resolve inflammation and promote tissue repair.
In diabetic wounds, this transition does not occur properly. Macrophages often remain stuck in a pro-inflammatory state, which prolongs inflammation and promotes tissue damage rather than healing.